Abstract
Many foods and food additives that are safe for human consumption can be extremely toxic to pets. Recognizing the clinical signs and clinicopathologic changes associated with these toxins allows prompt initiation of appropriate therapy. As with many other toxins, decontamination and supportive care are the mainstays of therapy for food toxicosis. Educating owners about foods and food additives that are unsafe for dogs and cats can help prevent toxicosis.
Some foods or food additives that are safe for humans can be toxic to dogs and cats. These include onions, grapes, foods containing xylitol, chocolate, and macadamia nuts. Large gaps remain in public knowledge pertaining to the risks of these potential toxins. As a result, many patients may not present for evaluation until after clinical signs have developed. It is important for clinicians to recognize these toxins as possible causes of a patient’s clinical signs to more thoroughly question the owner about exposure, to direct diagnostic testing and therapy, and to give a more accurate prognosis.
Garlic and Onions
Onions and garlic are members of the genus Allium, which also includes leeks, chives, and shallots. The toxicity of onions in dogs was first recognized in 1930, when dogs fed onions developed anemia during a research study.1 It has since been determined that onion juice, as well as dehydrated, powdered, and cooked onions, can cause hemolytic anemia in dogs and cats.2 Most cases of toxicosis are attributed to a single episode of accidental ingestion of raw onion or feeding of foods containing onions or garlic. The toxic dose has been found to be as low as 5 g/kg in cats and 15 g/kg in dogs.2 A medium-sized onion (2 to 3¼” in diameter) is approximately 150 g, and the average weight of a garlic clove is 6 g. A 10-lb dog would ingest a potentially toxic dose by eating only half of an onion. Crushing or chewing Allium spp releases allicin, which quickly breaks down into the toxic component n-propyl disulfide. N-propyl disulfide causes oxidative damage to hemoglobin and erythrocyte membranes, resulting in the formation of methemoglobin, Heinz bodies, and eccentrocytes, which then lead to intravascular hemolysis and anemia.3,4 Cats may be more sensitive to the toxic effects of onions because of differences in feline hemoglobin that result in an increased sensitivity to oxidative damage.
Clinical Signs
Initial signs of ingestion may include vomiting, diarrhea, and hypersalivation.2,3 Clinical signs may not develop for 1 to 5 days as hematologic changes begin, resulting in pale mucous membranes, weakness, ataxia, anorexia, depression, tachypnea, and tachycardia secondary to anemia.2 Icterus and hemoglobinuria may be seen secondary to intravascular hemolysis.2
Laboratory Findings
Abnormalities seen on a complete blood count (CBC) include decreases in packed cell volume (PCV), hematocrit, and hemoglobin concentration.2–4 Increased reticulocyte counts and neutrophilic leukocytosis may be seen in response to the anemia.3,4 Methemoglobin concentrations and the percentages of erythrocytes with Heinz bodies or classified as eccentrocytes are increased. 3,4 Hyperbilirubinemia is a common finding on a chemistry panel.2 Hemoglobinuria is seen on urinalysis and may result in an increased blood urea nitrogen level caused by transient nephrosis.2
Treatment
There is no specific antidote for onion or garlic toxicosis. If the patient presents within a few hours of ingestion of a potentially toxic amount, decontamination by emesis induction is recommended. Administration of activated charcoal with a cathartic may be beneficial if emesis is unsuccessful. Once clinical signs are present, treatment is supportive care. Intravenous (IV) fluids are recommended to correct dehydration from vomiting and promote diuresis to reduce the potential for hemoglobin nephrosis.2,5 The PCV should be monitored daily for 5 to 7 days. If anemia becomes severe, transfusions with whole blood or packed red blood cells may be needed.2,5 Antioxidants have not been shown to be beneficial.2
Prognosis
The prognosis depends on the severity of the anemia but is typically good with appropriate supportive care.2 Dogs with experimentally induced toxicosis eventually had complete resolution of anemia with no treatment.1,3,4
Grapes and Raisins
Grapes belong to the genus Vitis and are the largest crop fruit in the world.6 Since the mid-1990s, there have been numerous reports of acute renal failure (ARF) in dogs following ingestion of grapes, raisins, and Zante currants (black Corinth grapes).7 True currants are a berry of the genus Ribes, for which there are no published reports of toxicosis in dogs. There have been no definitive cases of raisin or grape toxicosis in cats, but some early reports suggest that a similar toxic reaction is possible in cats.7
At this time, the toxin and the mechanism of toxicity have not been determined. Several nephrotoxic agents, including herbicides, chlorinated hydrocarbons, carbamate, organophosphate insecticides, mycotoxins, and ochratoxin A, have been tested for but not found. These are now considered unlikely causes of kidney failure in cases of grape toxicosis.7 Not all dogs that ingest grapes or raisins develop ARF, which has led to a second theory that some dogs have an idiosyncratic reaction, resulting in renal ischemia secondary to hypovolemic shock.6,7 In dogs that do develop ARF, the lowest toxic doses reported are 19.6 g/kg (0.7 oz/kg) of grapes and 2.8 g/kg (0.1 oz/kg) of raisins.6 The ingested dose does not appear to correlate with the degree of renal damage.6,8
Clinical Signs
The earliest and most consistent clinical sign is vomiting, which typically begins within 24 hours of grape or raisin ingestion.6,7 Other clinical signs may include lethargy, anorexia, and diarrhea.7 As renal failure develops, abdominal pain and decreased urine output may be noted.8
Laboratory Findings
Chemistry findings are consistent with kidney failure, including azotemia, hypercalcemia, hyperkalemia, hyperphosphatemia, and an elevated calcium-phosphorus product.6–8 Elevations in alanine aminotransferase (ALT) and amylase activities have also been reported.8 Urinalysis results reflect proximal renal tubular damage resulting in isosthenuria or hyposthenuria, proteinuria, and glucosuria.6
Treatment
Because the development of ARF appears to be idiopathic, any dog with a history of grape or raisin ingestion should be treated immediately with gastric decontamination via emesis induction and the administration of activated charcoal with a cathartic.6–8 IV fluids should be initiated to promote diuresis and continued for a minimum of 48 hours, and renal values should be monitored for 72 hours.7 Treatment for renal failure includes IV fluids, histamine H2-receptor antagonists, and placement of a urinary catheter with closed collection system for early identification of oliguria or anuria. Treatment for oliguric or anuric renal failure can include aggressive therapy with IV fluids, furosemide, mannitol, peritoneal dialysis, and hemodialysis. Patients should be monitored closely for potential overhydration, which can result in hypokalemia, peritoneal effusion, pulmonary edema, and splenic congestion.6,8
Prognosis
For dogs that do not develop signs of toxicosis, the prognosis is excellent. Poor prognostic indicators include ataxia, weakness, metabolic acidosis, an elevated calcium-phosphorus product, hyperkalemia, oliguria, and anuria.6,7 In one study, for dogs that survived ARF secondary to grape or raisin toxicity, the average length of hospitalization was 6 days.6
Chocolate and Caffeine (Methylxanthines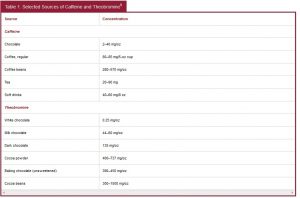 )
)
Methylxanthines, which are plant alkaloids, include theobromine, caffeine, and theophylline. While methylxanthines can be found in a variety of products, the most common toxicoses in dogs and cats result from the ingestion of chocolate, coffee beans, caffeine tablets, or cacao bean mulch.9 Other sources of caffeine in the kitchen include tea, coffee grounds, sodas, a variety of energy drinks, and products containing guarana. The median lethal dose (LD50) of caffeine in dogs is 140 mg/kg, while the LD50 of theobromine is 250 to 500 mg/kg.9 Cats are slightly more sensitive to methylxanthines, but these toxicoses are much rarer in cats because of their more discriminating eating habits. Concentrations of theobromine can range from 40 mg/oz in milk chocolate to >700 mg/oz in cocoa powder (), resulting in a potentially lethal dose of only 2oz/kg of milk chocolate or 0.2 oz/kg of baking chocolate.9,10
Clinical Signs
In dogs, caffeine is rapidly absorbed after ingestion, reaching peak serum levels within 60 minutes.9 Theobromine is absorbed 10 times more slowly, and the plasma half-life is more than 17 hours.9 Initial signs of toxicosis can be seen as early as 1 to 2 hours after ingestion and may include hyperactivity, vomiting, panting, ataxia, urinary incontinence, diarrhea, muscle tremors, tachycardia, dehydration, and hyperthermia.9,11 Signs may progress to hypertension, cardiac arrhythmias, muscle rigidity, seizures, respiratory failure, coma, and death.9 The severity of clinical signs is dose dependent, with 20 mg/kg resulting in mild signs, 40 to 50 mg/kg causing moderate to severe signs, and 60 mg/kg resulting in severe signs and seizures.10
Laboratory Findings
Caffeine commonly causes hypokalemia, and results of blood work may indicate dehydration.9 Theophylline commonly causes decreased serum potassium, phosphorus, and magnesium levels; hyperglycemia; mild metabolic acidosis; and leukocytosis.9 Electrocardiography (ECG) findings typically include tachycardia, but additional arrhythmias and premature ventricular contractions may be seen with more severe toxicosis.9
Treatment
Activated charcoal readily binds methylxanthines and should be administered initially with a cathartic. Because methylxanthines undergo enterohepatic recirculation and theobromine has a long plasma half-life, repeated doses of activated charcoal every 3 to 8 hours for up to 3 days may be beneficial for animals that have ingested very large doses or have ongoing clinical signs of toxicosis.9,11 The initial dose of activated charcoal with a cathartic should be 2 to 6 g/kg. Repeated doses should be smaller (0.5 g/kg) and administered without a cathartic. Patients should be monitored closely for the development of hypernatremia secondary to charcoal administration.9 IV fluids promote urinary excretion. Urinary catheterization or frequent walks may be beneficial to reduce reabsorption of unchanged caffeine from the urinary bladder, which could result in prolonged toxicosis.9,11 Benzodiazepines, barbiturates, or general anesthetics may be needed to control seizures and hyperactivity.9,12 Continuous ECG monitoring is recommended to detect significant arrhythmias. β-Blockers, such as propranolol (0.02 to 0.06 mg/kg IV slowly) or metoprolol (0.04 to 0.06 mg/kg IV q8h), may be used to control supraventricular tachycardia in dogs and cats.13 Ventricular arrhythmias resulting in hemodynamic instability in dogs are typically treated with lidocaine. Lidocaine is given at an initial loading dose of 2 to 4 mg/kg over 60 seconds followed by a constant-rate infusion (CRI) of 40 to 80 µg/kg/min.14 Lidocaine is not recommended in cats because of their increased sensitivity to its central nervous system (CNS) and cardiodepressant effects.11,14
Prognosis
The prognosis is typically excellent in animals that receive early decontamination and develop only mild to moderate signs of toxicosis. Animals with severe toxicosis that develop seizures and arrhythmias have a guarded prognosis.9
Alcohol and Bread Dough
Fermentation of sugars by certain species of yeast results in the production of ethanol. Ethanol is a two-carbon alcohol also known as ethyl alcohol, grain alcohol, or simply as spirits. Ethanol is found in a variety of products, such as solvents, fuels, paints, medications, and alcoholic beverages. In veterinary medicine, ethanol is commonly used to treat ethylene glycol (antifreeze) toxicosis. Ethanol toxicosis in animals most commonly results from accidental or intentional ingestion of alcoholic beverages. There are reports of alcohol poisoning in dogs after the ingestion of unbaked bread dough or rotting apples.15,16 The internal body temperature allows the yeast to begin the fermentation process, resulting in the production of carbon dioxide and increasing amounts of ethanol, which is then rapidly absorbed from the stomach.15
Clinical Signs
For dogs ingesting ethanol, the lowest lethal dose is 5.5 g/kg (5.5 mL/kg of 100% ethanol).17 Dividing the alcohol proof value by two gives the percentage of ethanol in the beverage.17 Ethanol is rapidly absorbed from the intestinal tract and crosses the blood-brain barrier, where it is believed to inhibit N-methyl-d-aspartate receptors.17 Most ethanol is metabolized in the liver by alcohol dehydrogenase and eliminated in the urine.15,17 Animals may have an initial period of vomiting and excitation followed by ataxia, lethargy, hypothermia, urinary incontinence, collapse, and CNS depression.18,19 Severe intoxication may result in coma, respiratory depression, cardiac arrest, seizures, and death.15,17–19 Dogs that have ingested bread dough may present with a distended, painful abdomen (from gas production), gastric obstruction, and the potential for gastric dilatation-volvulus.15
Laboratory Findings
Electrolyte derangements and hypoglycemia may be seen on a chemistry panel.15,19 Metabolic acidosis and respiratory acidosis are known to develop with ethanol toxicity.15,19 Measuring blood ethanol levels may be beneficial for determining prognosis and evaluating response to therapy.15,17 Adult dogs with a blood alcohol level of 1 mg/mL may show ataxia; blood levels of >4mg/mL are typically associated with coma.15 Local human hospitals are usually able to determine blood ethanol levels.
Treatment
Emesis should be induced with extreme caution and is recommended only for asymptomatic animals that have very recently ingested ethanol or bread dough.15,17 For dogs that have ingested bread dough, if emesis is unproductive, gastric lavage with cold water may be beneficial to slow fermentation, slow the production of alcohol, and, possibly, remove some of the dough.15 Treatment of alcohol toxicosis is mostly supportive, with administration of IV fluids to correct dehydration and enhance renal excretion. Monitoring parameters should include body temperature to prevent or correct hypothermia, blood gases for indications of respiratory or metabolic acidosis, and blood glucose; patients should also be monitored for seizures.15,17,19 Comatose patients with severe respiratory depression and respiratory acidosis require intubation and ventilation. Severe metabolic acidosis may require administration of sodium bicarbonate. For patients with severe toxicosis or prolonged recovery, hemodialysis has been shown to be beneficial for the rapid removal of ethanol.19
Prognosis
For mild cases of toxicosis and bread dough ingestion that are treated promptly, prognosis is excellent. Typically, with supportive care, clinical signs completely resolve within 24 hours.15,17 Animals with severe toxicosis resulting in respiratory depression and metabolic acidosis have a guarded prognosis.17
Xylitol
Xylitol is a five-carbon sugar alcohol that is found naturally in low concentrations in fruits, vegetables, berries, and mushrooms. Commercially produced xylitol is used in a variety of products, including sugar-free candy and gum, desserts, cereals, beverages, and toothpaste, and it comes in a powdered form for baking and cooking.20,21 While xylitol has a slow absorption and minimal effect on insulin levels in humans, it has been found to be profoundly toxic in dogs. Peak plasma levels of xylitol typically are reached within 30 minutes of ingestion, causing a significantly increased insulin level compared with an equivalent dose of glucose; this rapid increase in insulin may lead to severe hypoglycemia.20 Hepatotoxicity and fulminant hepatic failure have also been reported in dogs after ingestion of larger quantities of xylitol.21,22 The toxic dose of xylitol is considered to be >0.1g/kg for hypoglycemia and >0.5 g/kg for risk of hepatotoxicity. The severity of hepatotoxicity may be idiosyncratic rather than dose dependent because not all dogs that have ingested well above 0.5 g/kg have developed liver failure.20
Clinical Signs
Dogs commonly vomit after ingestion of xylitol. With ingestion of smaller toxic amounts, hypoglycemia caused by insulin release can develop within 30 to 60 minutes. Clinical signs of hypoglycemia include weakness, ataxia, recumbency, dull or altered mentation, altered vision, and seizures. With hepatotoxic doses, an initial period of hypoglycemia may not be seen or hypoglycemia may develop 24 to 48 hours after ingestion. Delayed hypoglycemia has been attributed to hepatic failure rather than xylitol’s direct stimulation of insulin secretion.22 As hepatic failure progresses and coagulopathy develops, additional clinical signs may include icterus, petechiae and ecchymoses on the skin and mucous membranes, hematochezia, and hematoma formation at venipuncture sites.22 The signs of acute hepatic failure may develop 2 to 72 hours after ingestion.21,22
Laboratory Findings
Hypoglycemia is the most common initial abnormality. Occasionally, hyperglycemia has been reported. This may be attributed to the Somogyi phenomenon, in which profound hypoglycemia in response to excess insulin stimulates glucagon, epinephrine, cortisol, and growth hormone, resulting in rebound hyperglycemia.20 Hypokalemia and hypophosphatemia are also common in cases of low-dose xylitol ingestion. Hyperphosphatemia may develop with liver failure and may be associated with increased mortality.20 Other abnormalities associated with hepatic failure include prolonged clotting times, thrombocytopenia, increased ALT and alkaline phosphatase (ALP) activities, and hyperbilirubinemia.20
Treatment
When possible, the dose of xylitol ingested should be determined to guide treatment recommendations. To estimate the amount of xylitol ingested, consider 0.3 g/piece of gum if xylitol is not the first ingredient listed (for gum) and 190 g/cup as an estimate for xylitol in baked goods. If the total sugar alcohol content is labeled and xylitol is the first ingredient listed, the xylitol content should be estimated as the total sugar alcohol content.20 Induction of emesis should only be attempted if hypoglycemia is not already present and a large dose has been ingested.
Evidence suggests that activated charcoal does not significantly bind xylitol; however, it may be beneficial if given with a cathartic in cases of acute, large-dose ingestion.20,23 Dogs ingesting between 0.1 and 0.5 g/kg of xylitol should be monitored for the development of hypoglycemia every 1 to 2 hours for at least 12 hours.20 If hypoglycemia is develops, 1 mL/kg of 50% dextrose diluted 1:2 with a crystalloid solution should be administered IV followed by a CRI of 2.5% to 5% dextrose to maintain a normal blood glucose level.20 A CRI of dextrose should be started immediately in any dog that has ingested >0.5 g/kg of xylitol, even if the blood glucose is normal at presentation.20 Electrolytes and phosphorus levels should be monitored every 4 to 6 hours with the addition of potassium and phosphorus supplementation as needed. Liver enzymes, total bilirubin level, platelet count, erythrocyte count, and coagulation times should be evaluated on presentation and thereafter monitored every 24 hours for 72 hours.20 For dogs that present in liver failure or that ingested a potentially hepatotoxic dose, liver protectants and antioxidants, such as s-adenosyl-methionine (SAMe), silymarin, N-acetylcysteine, and vitamin E, may be beneficial.20 Frozen or fresh frozen plasma should be administered in patients with active hemorrhage caused by coagulopathies.
Prognosis
For animals that develop only uncomplicated hypoglycemia controlled with IV dextrose, the prognosis is good. The prognosis is guarded to poor for dogs that develop hepatotoxicosis and hepatic failure.20
Macadamia Nuts
Macadamia nuts are harvested primarily in Hawaii from the Macadamia integrifolia and Macadamia tetraphylla trees.24,25 Macadamia nut toxicity has been reported only in dogs.24 It is unknown if the toxic component is in the nuts themselves, a processing contaminant, or a mycotoxin associated with the nuts.24–26 The minimum reported dose that has resulted in clinical signs is 2.2 g/kg.24,26
Clinical Signs
Clinical signs generally develop within 6 to 12 hours after ingestion, although there have been reports of clinical signs as early as 60 minutes after ingestion.25,26 Initial clinical signs most frequently include weakness, depression, vomiting, ataxia, tremors, and hyperthermia.24,26 Signs can progress to include CNS depression, stiffness, abdominal pain, and significant hindlimb weakness resulting in recumbency.24 Once assisted into a standing position, dogs can typically walk and have normal peripheral reflexes but diminished or absent conscious proprioception.24
Laboratory Findings
Abnormal findings on a chemistry panel include marked increases in serum lipase, moderately increased serum triglycerides, and mildly elevated serum ALP.25,26 The lipase values commonly return to normal within 48 hours, but ALP may remain elevated.25 Abnormal CBC findings include elevated total white blood cell counts with a significant increase in segmented neutrophils.24 As with the chemistry values, CBC abnormalities typically return to normal within 48 hours.25
Treatment
If ingestion is recent and the dog is asymptomatic, emesis induction should be considered.25 Although the toxic component has not been identified, administration of a single dose of activated charcoal with a cathartic has been recommended to hasten passage of the nuts through the intestinal tract if a large dose was ingested.25,26 Most cases of uncomplicated, clearly evident macadamia nut toxicosis can be managed with symptomatic care at home.25,26 Very young, older, or more debilitated dogs, or dogs with preexisting medical conditions may require hospitalization for monitoring and IV fluids therapy if clinical signs are moderate to severe.25 Additional treatment for methylxanthine toxicity may be required if the nuts were chocolate coated or in brownies. The amount of chocolate ingested should be calculated and treatment should be based on the recommendations outlined above for methylxanthine toxicosis.
Prognosis
Potential complications may include pancreatitis, gastroenteritis, or intestinal obstruction.24 In most cases, the prognosis is excellent, with most dogs returning to normal within 24 to 48 hours.26
Conclusions
Obtaining a detailed history, recognizing the clinical signs, understanding the clinicopathologic changes, and rapidly initiating appropriate therapy can greatly improve the prognosis for pets exposed to these potentially toxic foods. Preventing exposure is the key to reducing the incidence of food toxicosis. Veterinarians and technicians play a vital role in educating pet owners about the dangers of these common foods.
References
1. Sebrell WH. An anemia in dogs produced by feeding onions. Public Health Rep 1930;45:1175-1189.
2. Cope RB. Allium species poisoning in dogs and cats. Vet Med 2005;100(8):562-566.
3. Harvey JW, Rackear D. Experimental onion-induced hemolytic anemia in dogs. Vet Pathol 1985;22:387-392.
4. Lee K, Yamato O, Tajima M, et al. Hematologic changes associated with the appearance of eccentrocytes after intragastric administration of garlic extract to dogs. Am J of Vet Res 2000;61:1446-1450.
5. Osweiler GD. Plant-related toxicosis. In: Nieginski EA, ed. The National Veterinary Medical Series: Toxicology. Philadelphia: Lippincott Williams and Wilkins; 1996:361-403.
6. Eubig PA, Brady MS, Gwaltney-Brant SM, et al. Acute renal failure in dogs after the ingestion of grapes or raisins: a retrospective evaluation of 43 dogs (1992-2002). J Vet Intern Med 2005; 19(5):663-674.
7. Mostrom MS. Grapes and raisins. In: Peterson ME, Talcott PA, eds. Small Animal Toxicology. 2nd ed. St. Louis: Elsevier Saunders; 2006:727-731.
8. Mazzaferro EM, Eubig PA, Hackett TB, et al. Acute renal failure associated with raisin or grape ingestion in 4 dogs. J Vet Emerg Crit Care 2004;14(3):2003-212.
9. Carson TL. Methylxanthines. In: Peterson ME, Talcott PA, eds. Small Animal Toxicology. 2nd ed. St. Louis: Elsevier Saunders; 2006:845-852.
10. Gwaltney-Brant S. Chocolate intoxication. Vet Med 2001;96(2):108-111.
11. Luiz JA, Heseltine J. Five common toxins ingested by dogs and cats. Compend Contin Educ Pract Vet 2008;30(11):578-587.
12. Plumb DC. Diazepam. In: Plumb DC, ed. Veterinary Drug Handbook. 7th ed. Ames, IA: Wiley-Blackwell; 2011:408-414.
13. Beasley V, Dorman D, Fikes J, et al. Chocolate, caffeine, and other methylxanthines. In: Beasley V, ed. A Systems Affected Approach to Veterinary Toxicology. St. Louis: Mosby; 1999:116-120.
14. Plumb DC. Lidocaine HCL. In: Plumb DC, ed. Veterinary Drug Handbook. 7th ed. Ames, IA: Wiley-Blackwell; 2011:805-809.
15. Means C. Bread dough toxicosis in dogs.J Vet Emerg Crit Care 2003;13(1):39-41.
16. Kammerer M, Sachot E, Blanchot D. Ethanol toxicosis from the ingestion of rotten apples by a dog. Vet Hum Toxicol 2001;43(6):349-350.
17. Richardson JA. Ethanol. In: Peterson ME, Talcott PA, eds. Small Animal Toxicology. 2nd ed. St. Louis: Elsevier Saunders; 2006:698-701.
18. Houston DM, Head LL. Acute alcohol intoxication in a dog. Can Vet J 1993;34:41-42.
19. Keno LA, Langston CE. Treatment of accidental ethanol intoxication with hemodialysis in a dog. J Vet Emerg Crit Care 2011;21(4):363-368.
20. Piscitelli CM, Dunayer EK, Aumann M. Xylitol toxicity in dogs. Compend Contin Educ Pract Vet 2010;32(2):E1-E4.
21. Todd JM, Powell LL. Xylitol intoxication associated with fulminant hepatic failure in a dog. J Vet Emerg Crit Care 2007;17(3):286-289.
22. Dunayer EK, Gwaltney-Brant SM. Acute hepatic failure and coagulopathy associated with xylitol ingestion in eight dogs. JAVMA 2006;229(7):1113-1117.
23. Cope RB. A screening study of xylitol binding in vitro to activated charcoal. Vet Hum Toxicol 2004;46(6):336-337.
24. Hansen SR, Buck WB, Meerdink G, et al. Weakness, tremors, and depression associated with macadamia nuts in dogs. Vet Hum Toxicol 2000;42:18-21.
25. Gwaltney-Brant SM. Macadamia nuts. In: Peterson ME, Talcott PA, ed. Small Animal Toxicology. 2nd ed. St. Louis: Elsevier Saunders;2006:817-821.
26. Hansen SR. Macadamia nut toxicosis in dogs. Vet Med 2002;97:275-276.




